Axitec Co., Ltd
Company Details
Company Introduction
Axitec is established on the fundamental principles of enhancing the value and competitiveness of the business entities that it serves. At Axitec, we work hard with a single purpose of delivering excellent outcomes to the customers.The company commenced operations in Shen Zhen, China, in December 2003. We have solid-standing expertise in the process development, scale-up and cGMP production of a variety of generic APIs and advanced pharmaceutical intermediates. The experience of our R&D scientists, the capabilities of our chemists, the flexibility of our facilities and the competence of our analytical support enable us to provide to our customers products of a global quality. Equally important, we are familiar with European and U.S. Intellectual Property Rights.
Company Information
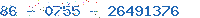
Related Company
- Two Klemm
- Handan Zhengxin Fastener Co. ,Ltd
- Yilimed Medical Supplies Inc
- Wonbiogen Co., Ltd.
- Shenzhen Zonecent Tehchnologies Co., Ltd.
- WODA Technology LIMITED
- Beijing Shangjia Equipment Co., Ltd.
- Shenzhen City Yuncheng Plate-Making Co., Ltd.
- Eyelpoker
- Bino Scientific
- Oriental Trade Concer
- Guangzhou Lanbolight Techology Co.,Ltd